|
|
|
This year, an estimated 1.4 million Americans will have a new or recurrent heart attack.
An identified risk factor for osteoporosis is the intake of excessive amounts of vitamin A. Dietary intake of approximately double the recommended daily amount of vitamin A, by women, has been shown to reduce bone mineral density and increase the chances for hip fractures compared with women who consumed the recommended daily amount (or less) of vitamin A.
To combat osteoporosis, changes in lifestyle and diet are recommended. At-risk patients should include 1,200 to 1,500 mg of calcium daily either via dietary means or with supplements.
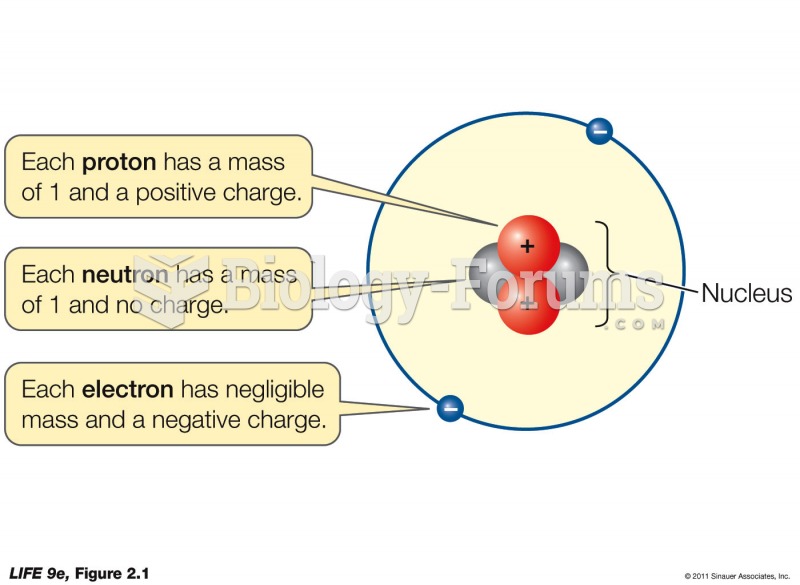
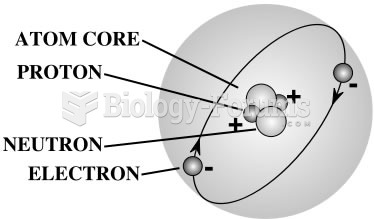
The ratio of hydrogen atoms to oxygen in water (H2O) is 2:1.
Nearly 31 million adults in America have a total cholesterol level that is more than 240 mg per dL.