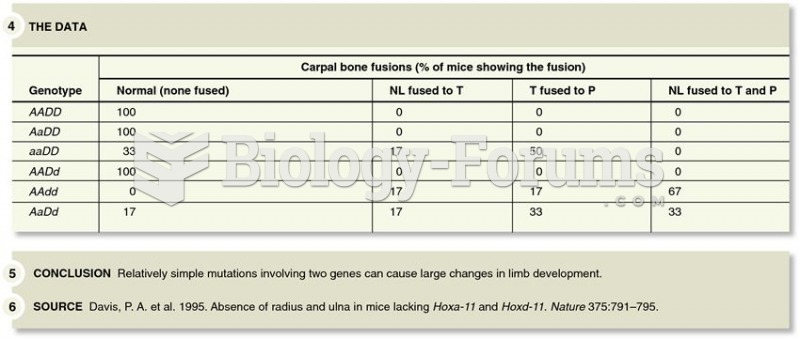
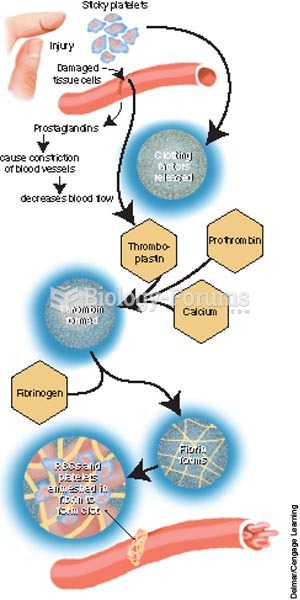
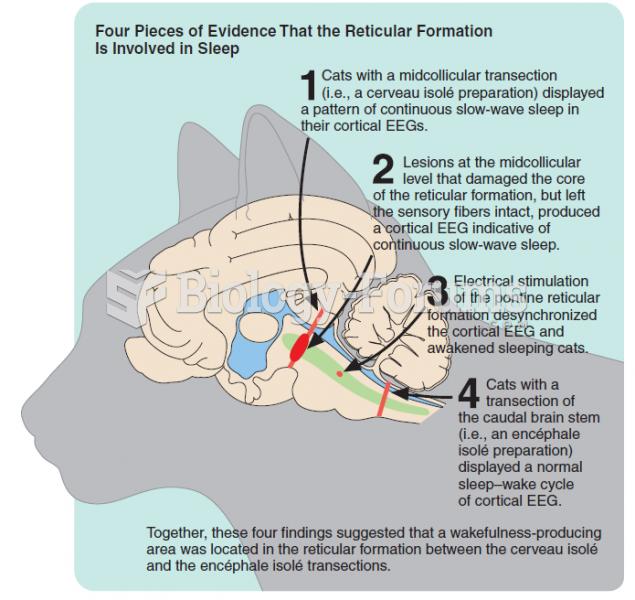
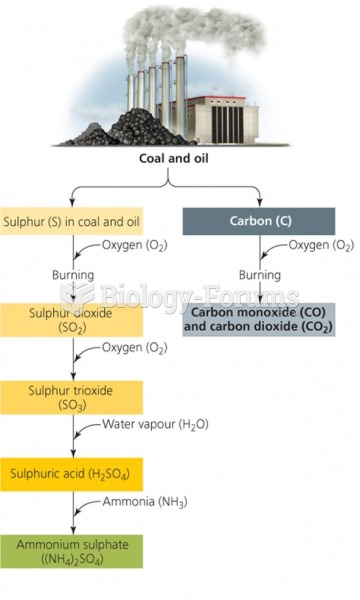
|
|
|
There are more sensory neurons in the tongue than in any other part of the body.
Bacteria have been found alive in a lake buried one half mile under ice in Antarctica.
After 5 years of being diagnosed with rheumatoid arthritis, one every three patients will no longer be able to work.
Many of the drugs used by neuroscientists are derived from toxic plants and venomous animals (such as snakes, spiders, snails, and puffer fish).
There are major differences in the metabolism of morphine and the illegal drug heroin. Morphine mostly produces its CNS effects through m-receptors, and at k- and d-receptors. Heroin has a slight affinity for opiate receptors. Most of its actions are due to metabolism to active metabolites (6-acetylmorphine, morphine, and morphine-6-glucuronide).