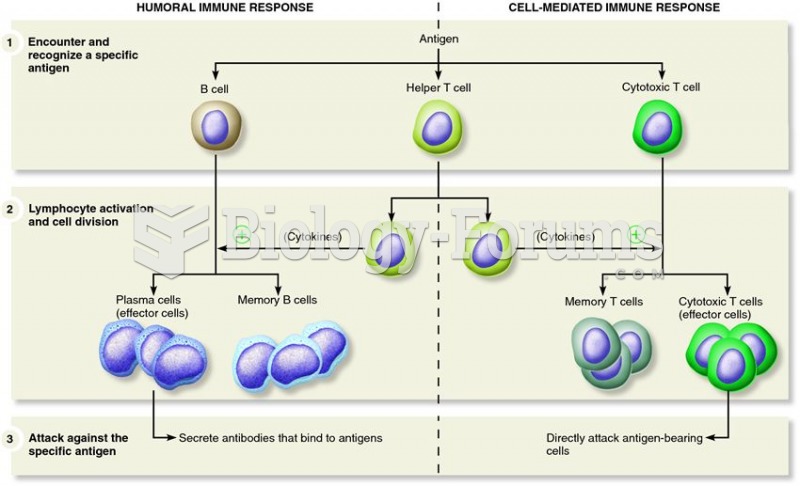
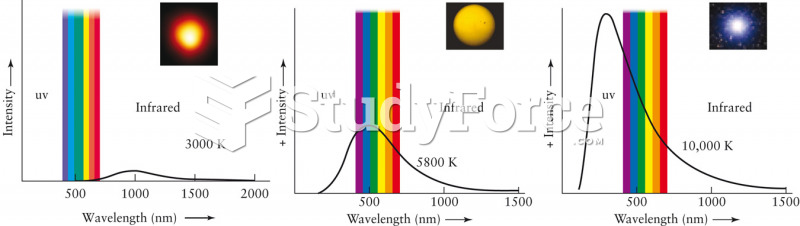
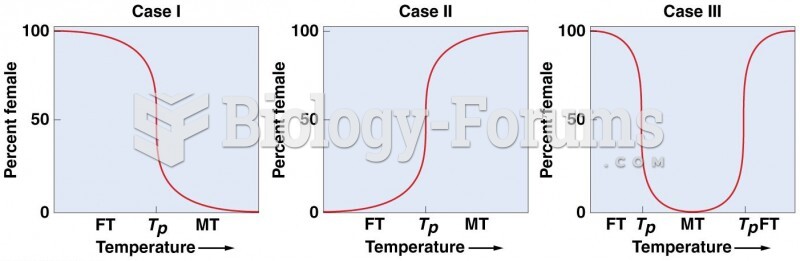
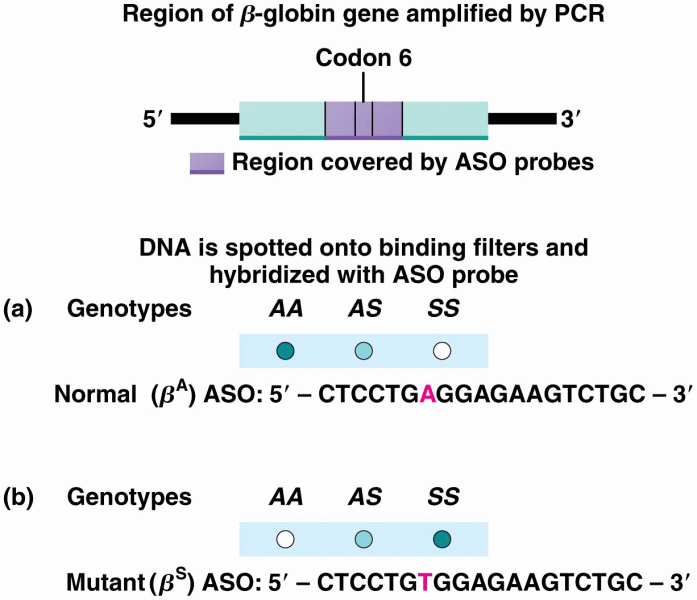
|
|
|
The term bacteria was devised in the 19th century by German biologist Ferdinand Cohn. He based it on the Greek word "bakterion" meaning a small rod or staff. Cohn is considered to be the father of modern bacteriology.
When blood is exposed to air, it clots. Heparin allows the blood to come in direct contact with air without clotting.
After 5 years of being diagnosed with rheumatoid arthritis, one every three patients will no longer be able to work.
Asthma cases in Americans are about 75% higher today than they were in 1980.
A cataract is a clouding of the eyes' natural lens. As we age, some clouding of the lens may occur. The first sign of a cataract is usually blurry vision. Although glasses and other visual aids may at first help a person with cataracts, surgery may become inevitable. Cataract surgery is very successful in restoring vision, and it is the most frequently performed surgery in the United States.