|
|
|
To combat osteoporosis, changes in lifestyle and diet are recommended. At-risk patients should include 1,200 to 1,500 mg of calcium daily either via dietary means or with supplements.
If you could remove all of your skin, it would weigh up to 5 pounds.
More than one-third of adult Americans are obese. Diseases that kill the largest number of people annually, such as heart disease, cancer, diabetes, stroke, and hypertension, can be attributed to diet.
Many people have small pouches in their colons that bulge outward through weak spots. Each pouch is called a diverticulum. About 10% of Americans older than age 40 years have diverticulosis, which, when the pouches become infected or inflamed, is called diverticulitis. The main cause of diverticular disease is a low-fiber diet.
In ancient Rome, many of the richer people in the population had lead-induced gout. The reason for this is unclear. Lead poisoning has also been linked to madness.
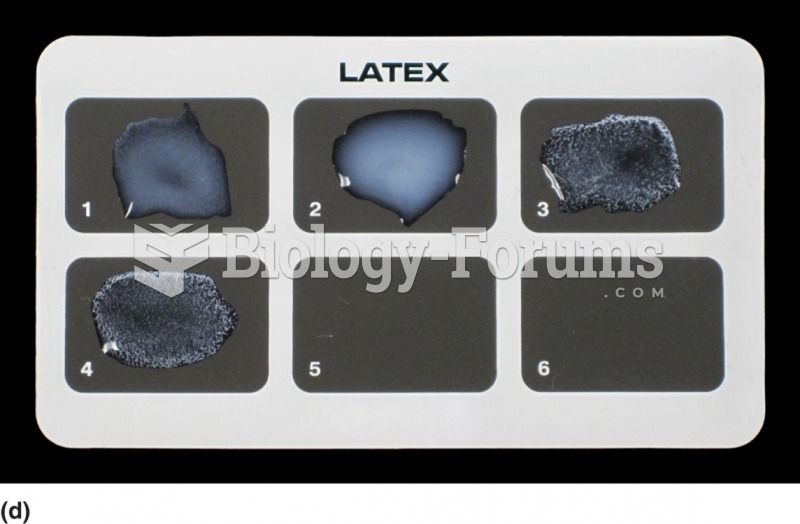 A latex agglutination reaction. This is an analysis for rheumatoid factor. Immunoglobulin is bound ...
A latex agglutination reaction. This is an analysis for rheumatoid factor. Immunoglobulin is bound ...
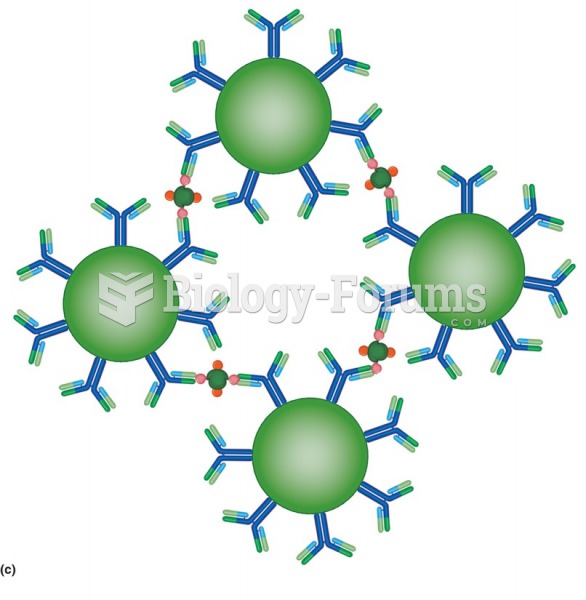
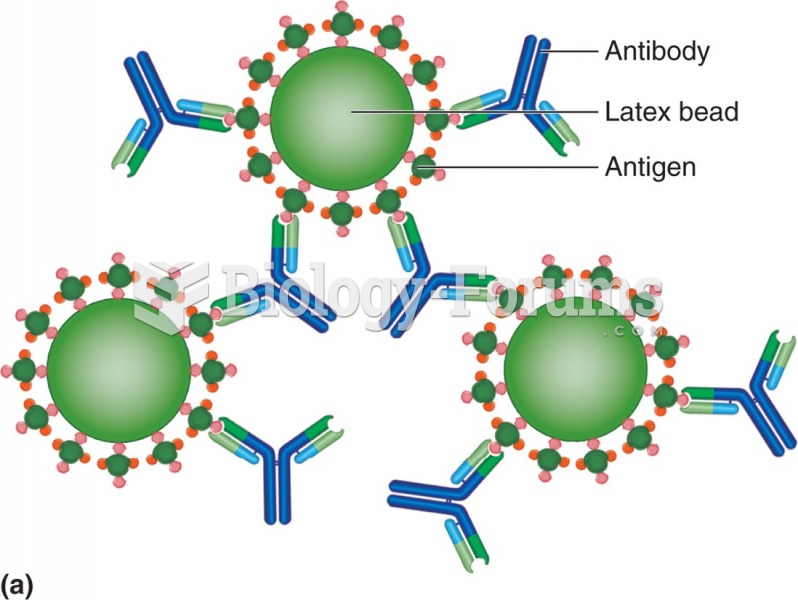 An agglutination reaction. Antigen-coated latex beads are held together by antibody. In a direct ...
An agglutination reaction. Antigen-coated latex beads are held together by antibody. In a direct ...