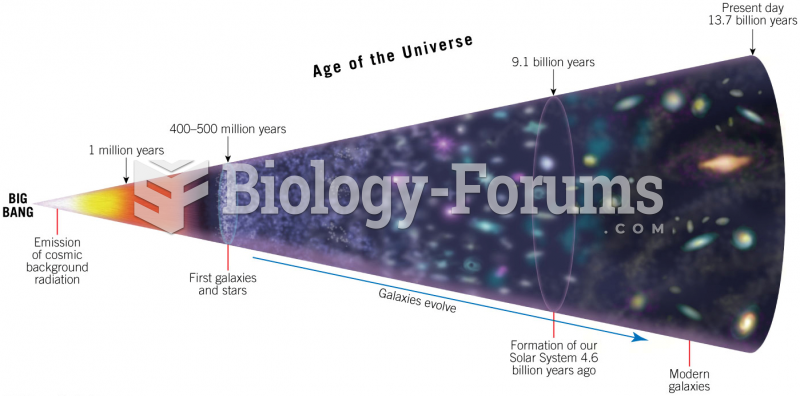
|
|
|
Your heart beats over 36 million times a year.
Side effects from substance abuse include nausea, dehydration, reduced productivitiy, and dependence. Though these effects usually worsen over time, the constant need for the substance often overcomes rational thinking.
Human neurons are so small that they require a microscope in order to be seen. However, some neurons can be up to 3 feet long, such as those that extend from the spinal cord to the toes.
Bisphosphonates were first developed in the nineteenth century. They were first investigated for use in disorders of bone metabolism in the 1960s. They are now used clinically for the treatment of osteoporosis, Paget's disease, bone metastasis, multiple myeloma, and other conditions that feature bone fragility.
The term bacteria was devised in the 19th century by German biologist Ferdinand Cohn. He based it on the Greek word "bakterion" meaning a small rod or staff. Cohn is considered to be the father of modern bacteriology.
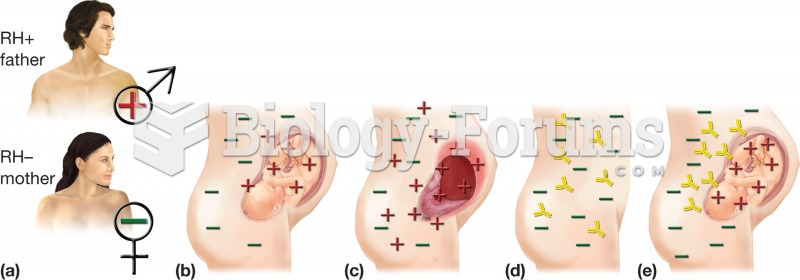 Erythroblastosis fetalis. (a) The condition occurs with an RH+ father and RH– mother. (b) First preg
Erythroblastosis fetalis. (a) The condition occurs with an RH+ father and RH– mother. (b) First preg
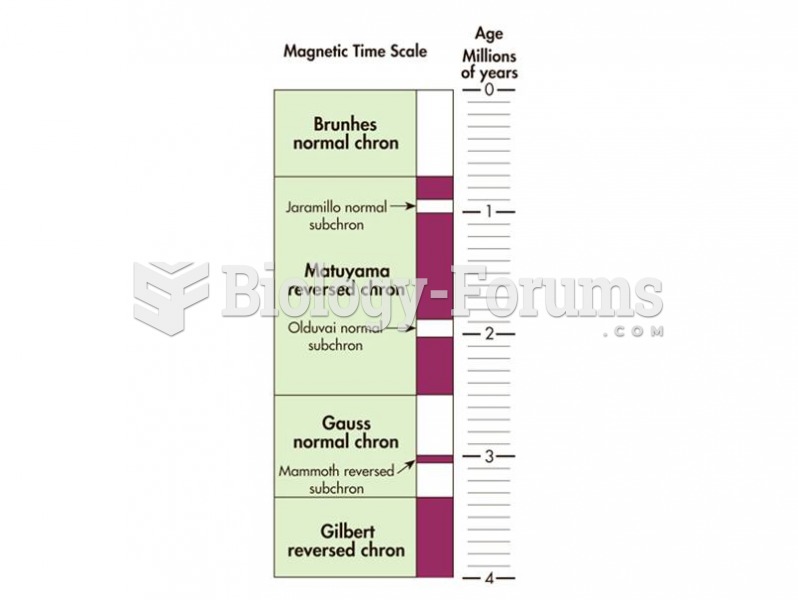 The geomagnetic polarity time scale shows how Earth’s magnetic pole has changed through geologic tim
The geomagnetic polarity time scale shows how Earth’s magnetic pole has changed through geologic tim